Understanding the concept of isotopes in its right essence is very important in the field of physics. Isotopes are majorly used in scientific applications, radioactive reactors, health sector and more fields that are relevant to human life. Here are the basic facts about isotopes and why the study of the isotope is important.
Defining Isotopes
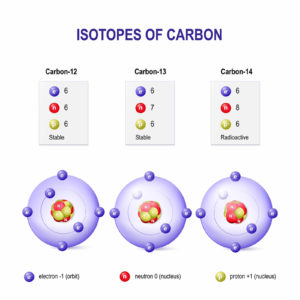
Isotopes are atoms with a same number of protons, but they differ in the number of neutrons. In other words, isotopes are atoms with the same atomic number, but different mass number. Scientifically, isotopes can be defined as the variants of a specific element, with the difference in the number of neutrons.
Understanding Isotopes
You will need to be clear about the structure of an atom, it constitutes of electrons, neutrons, and protons. The nucleus of the atom is comprised of protons and neutrons while the electrons revolve around the nucleus. Atomic mass is the sum of the number of neutrons and number of protons in the nucleus. In an isotope of any particular element, the number of protons will be same, but the number of neutrons will be different. The isotopes of the same element may show different properties.
Isotopes are classified into stable isotopes and radioactive isotopes. Stable isotopes have a stable combination of protons and neutrons while radioactive can be unstable which will undergo decay. During the process, it emits alpha, gamma or beta rays.
For example, one type of isotope of a particular element can be stable, while another isotope of the same element can be unstable or radioactive. Consider the example of hydrogen, which is one of the most abundant elements on the planet. Protium, deuterium, and tritium are isotopes of hydrogen. Protium’s neutron number is zero, while deuterium has a neutron, and the third isotope tritium has two neutrons.
Why Is Isotope Useful?
Isotopes were discovered by researchers around a century ago. It has been a leap for the scientific world with a wide variety of possible applications. According to scientists, there are around ninety elements that are naturally occurring with more than 250 isotopes. It also forms around 3200 other isotopes which are categorized as radioactive or unstable. Since isotopes of the same element can show different properties, scientists see a lot of possibilities in newer applications.
Mainly, isotopes are used in smoke detectors and in control rods in nuclear power reactors. Isotopes have been widely used in medical science, from the diagnosis of diseases using imaging techniques to the sterilization of medical equipment.
Make It Easy to Learn
Are you finding it difficult to learn physics and considering physics tuition? As the student gets individual attentions and extra coaching, our JC physics tuition would not only help to gain good score but also in understanding the complex concepts in the right way.

